The winter season in temperate areas is characterized by the mixing of water masses. This process ceases to exist when the heat balance between air and water becomes negative, hence contributing to the accumulation of phytoplankton in the proximity of the surface where a spring bloom takes place upon stratification of water masses. The abundance of phytoplanktonic species close to the surface limits the light availability for the lower situated photosynthetic organisms and induces them to synthetize pigments for a more efficient light capture. Within days to weeks, the phytoplankton found in the surface layer consume the nutrients, and thus become less abundant and sink to the base of the euphotic zone (~ 100 meters, although it may vary according to the area studied), where they have access to light from above and nutrients from below, called the deep chlorophyll maximum (DCM).
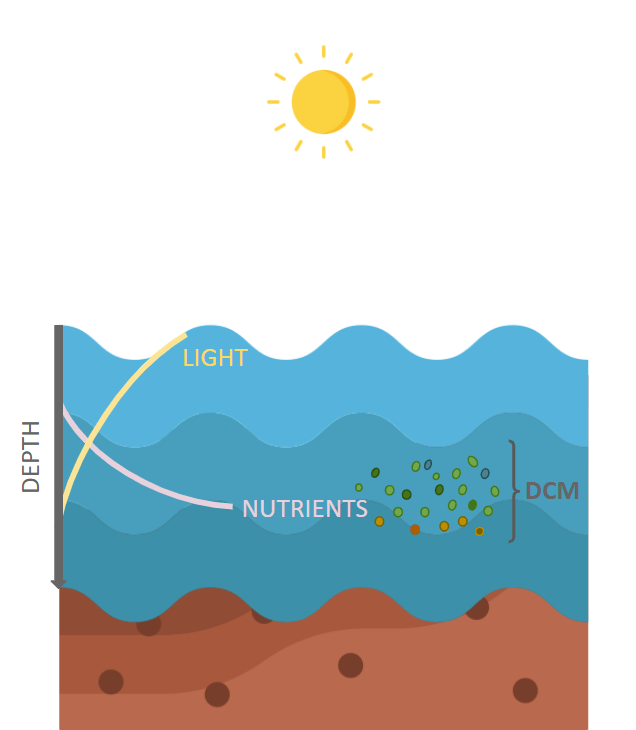
It was previously thought that this layer, once established, is quite stable and that the distribution of phytoplankton species within it is homogeneous. However, physical and biogeochemical processes, such as internal tides and rough topography that induce diapycnal mixing of nutrients, especially nitrate, towards upper DCM regions, are thought to influence the changes in the local community structure and sustain the production of organic matter in the oceans. Another physical process that has been discovered to impact the DCM is the presence of eddies, that are vortical circulations of water masses which involve vertical transport of nutrients upwards or downwards, depending on the direction of circulation. If an eddy is cyclonic, it circles in an anticlockwise direction in the Northern hemisphere, and the reverse is true for an anticyclonic eddy. In a cyclonic eddy, the force diverges the water masses creating space, therefore pulling up deep seawater to the euphotic zone in the center of the eddy. Consequently, the upwelling produced brings up nutrient resources, enhancing the primary production and increasing the biomass.
Different photosynthetic groups position heterogeneously within a DCM by searching their preferential ecological niche. Observations show that Prochlorococcus stays in the shallowest part of the DCM, followed by dinoflagellates, Synechococcus and small prymnesiophyte species. Whereas coccolithophores, chlorophytes, pelagophytes and diatoms prefer the deepest parts of the DCM. Considering that temperature and light availability decrease with depth, while the nutrient concentration increases, different phytoplankton groups acclimate accordingly to benefit the most from these peculiar conditions that are found within a DCM.
Prochlorococcus, for example, is known to possess the ability of living in low nutrient conditions, while dinoflagellates and Synechococcus normally lean towards more nutrient rich parts but easily acclimate to environments that lack nutrients. In the case of prymnesiophytes, they are believed to have mixotrophic abilities, meaning that they can either perform photosynthesis or switch to heterotrophy when the light becomes depleted. Although many questions remain to be answered in regards to the distribution of chlorophytes within the DCM, small pelagophytes were found to genetically modify to synthesize light-harvesting complexes in low light conditions and form cellular structures involved in nitrogen assimilation. Diatoms, on the other hand, having an efficient photosystem for low light, are prone to obtain nutrient pulses that come from intertidal oscillations and eddies.
These two perturbation processes require further research since they are intricately connected to both the ecosystem response and ocean warming fluctuations. How will global warming impact the vertical stratification on the global scale? Will it influence the nutrient mixing to the upper parts of the oceans? How will the primary production change? What oscillations, if any, will be observed in the biological pump? These are some questions that our and future generations will have to answer.
Huisman, J., Thi, N.N.P., Karl, D.M. and Sommeijer, B., 2006. Reduced mixing generates oscillations and chaos in the oceanic deep chlorophyll maximum. Nature, 439(7074), pp.322-325.
Latasa, M., Cabello, A.M., Morán, X.A.G., Massana, R. and Scharek, R., 2017. Distribution of phytoplankton groups within the deep chlorophyll maximum. Limnology and Oceanography, 62(2), pp.665-685.
Tuerena, R.E., Williams, R.G., Mahaffey, C., Vic, C., Green, J.M., Naveira‐Garabato, A., Forryan, A. and Sharples, J., 2019. Internal tides drive nutrient fluxes into the deep chlorophyll maximum over mid‐ocean ridges. Global Biogeochemical Cycles, 33(8), pp.995-1009.
Zhang, Y., Kieft, B., Hobson, B.W., Ryan, J.P., Barone, B., Preston, C.M., Roman, B., Raanan, B.Y., Marin III, R., O’Reilly, T.C. and Rueda, C.A., 2019. Autonomous tracking and sampling of the deep chlorophyll maximum layer in an open-ocean eddy by a long-range autonomous underwater vehicle. IEEE Journal of Oceanic Engineering, 45(4), pp.1308-1321.


One response to “The Deep Chlorophyll Maximum”
Bell´articolo 👌